Dr Alizadeh is the proud recipient of multiple awards including the prestigious Ellis island Medal of Honor.
A Top Specialist Doctor in New York
Dr. Kaveh Alizadeh, a top plastic surgeon in NYC and Long Island, is board-certified by the American Board of Plastic Surgery. He currently serves as the Professor of Clinical Surgery at New York Medical College and was formerly the Chief of Plastic and Reconstructive Surgery at Westchester Medical Center.
From 2007 to 2013, Dr. Alizadeh served as the president of the largest and oldest plastic surgery center in North America. He has served as the president of the New York Regional Society of Plastic Surgeons and was also elected as the president of the New York State Society of Plastic Surgeons where he has served on the board of trustees. He is also the oral examiner for the American Board of Plastic Surgery.
In 2014, Dr. Alizadeh opened his own private practice with a vision towards the future. He includes world class politicians, Oscar winning actors, and Grammy winning musicians among his many patients. He has been recognized as a Castle Connolly, US and World News Top Doctor for 20 consecutive years.

Continuous Contribution To The Field
Dr. Alizadeh directs the Clinical Research Division of Cosmoplastic Surgery, where he has conducted multiple national clinical trials in plastic surgery devices, breast surgery, and injectables for rejuvenation. He has served as primary clinical trial investigator for the FDA for various devices for surgical and non-surgical procedures.
He has published dozens of peer reviewed articles and given over 100 international lectures and presentations in his specialty. He is credited with developing a new breast-lift technique, NaturaBra®, as well as "progressive" eyelid-lift and abdominoplasty techniques.
Since 2005, he has been recognized as a Top Doctor by Consumer Research Council, and since 2010, a Castle Connolly Top Doctor. Dr. Alizadeh has also received national recognition as a Top Doctor by U.S. News and World Report. He has taught his techniques to hundreds of surgeons in 27 countries worldwide and provides free healthcare via his non-profit organization, Mission: Restore.

Media & Awards
Dr. Alizadeh has appeared in over 60 media outlets such as CNN, CBS 60 Minutes, NBC Today Show, Discovery Health, Newsweek, Wall Street Journal, and The New York Times. He has authored over 30 publications and has given extensive presentations at the national and international level. In 2009, Dr. Alizadeh's humanitarian volunteer work earned him the prestigious Ellis Island Medal of Honor, an award recorded in the United States Congressional Record.

Featured Podcast
Wide Range Of Elective Offerings
Dr. Alizadeh specializes in a range of cosmetic and reconstructive surgical procedures, as well as less-invasive treatments. Cosmetic procedures include breast augmentation, lift, and reduction; body contouring, including buttock augmentation and labiaplasty; as well as minimally invasive facial rejuvenation. Reconstructive procedures include migraine surgery and post-cancer breast reconstruction. He also performs the latest non-invasive facial rejuvenation techniques, including injectables such as RADIESSE® (+), Restylane® Silk, Restylane® Lyft, KYBELLA®, JUVÉDERM VOLUMA® XC, BOTOX® Cosmetic, XEOMIN®, Dysport®, JUVÉDERM®, and Sculptra® Aesthetic, for which he is a certified national trainer.
Dr. Kaveh Alizadeh shares the indivisible journey between his own hard childhood experiences and his calling to train doctors and save lives wherever there is conflict, war and trauma. To date, he has trained over 100 surgeons in plastic surgery. He is a sought-after motivational speaker because of his personal journey as a refugee, a doctor, and as a teacher. You can see a speech he delivered to over two thousand participants at the 2019 YPO global Leadership Conference in Cape Town, South Africa.
Schedule A Consultation
Contact UsLeaders Across Borders
Dr. Kaveh Alizadeh shares the indivisible journey between his own hard childhood experiences and his calling to train doctors and save lives wherever there is conflict, war and trauma. He is a sought-after motivational speaker because of his personal journey as a refugee, a doctor, and as a teacher. You can see a speech he delivered to over two thousand participants at the 2019 YPO global Leadership Conference in Cape Town, South Africa.

Training
Dr. Alizadeh earned his undergraduate degree from Cornell University and his Master's Degree from Columbia College of Physicians and Surgeons. He then received his MD from Cornell University Medical College, with commendation from the Dean. Dr. Alizadeh completed his General Surgery and Plastic Surgery training at the University of Chicago Hospitals, followed by an additional year of subspecialty training in Cosmetic Surgery, Microsurgery, and Breast Reconstruction at Memorial Sloan Kettering Cancer Center in New York. He has received further executive education training at Harvard Business School.
Gallery
View More
Education

- 2006-2007: Harvard Business School Executive Program
- 1990-1993: Cornell University Medical College; New York, New York – Doctor of Medicine, Joseph Collins Award
- 1988-1990: Columbia College of Physicians and Surgeons; New York, New York – Masters of Science
- 1986: Sorbonne University; Paris, France - Certificat de la Civilization Francaise
- 1984-1988: Cornell University; Ithaca, New York - Bachelor of Arts
- 1981-1984: Dwight Englewood School; Englewood Cliffs, New Jersey - Distinguished Alumnus Award 2005
Director of Cosmetic Surgery at Long Island, NY's Cosmoplastic Surgery
- 1999-2000: Fellowship: Microsurgery, Cosmetic Surgery, Memorial Sloan-Kettering Hospital, NY; Manhattan Eye, Ear and Throat Hospital, NY
- 1998-1999: Chief Resident: Plastic and Reconstructive Surgery, The University of Chicago Hospitals
- 1997-1999: Residency: Plastic and Reconstructive Surgery, The University of Chicago Hospitals
- 1994-1997: Residency: General Surgery, The University of Chicago Hospitals; Chicago

Positions




Professor of Surgery
New York Medical College
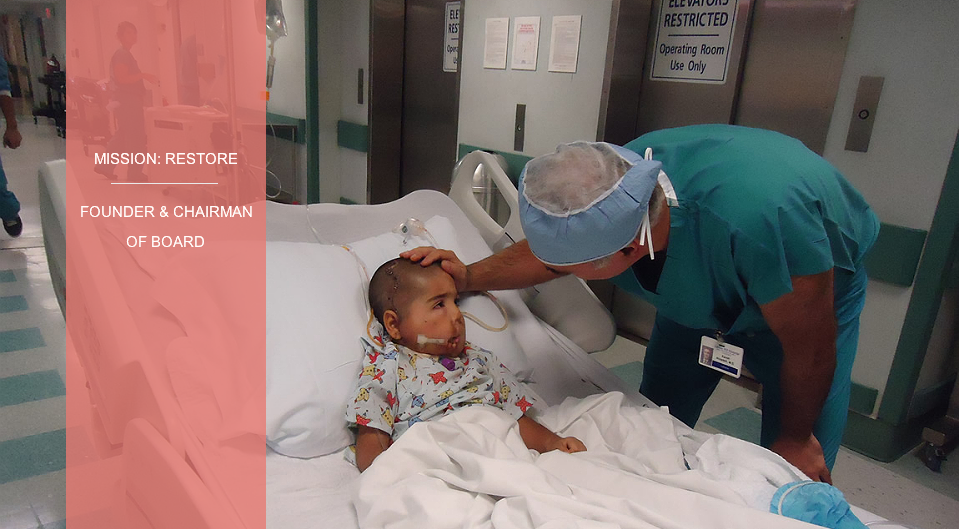
Founder and Chairman Of Board
Mission: Restore, which provides clinical services, education, and research for victims of war and trauma around the world
President
Long Island Plastic Surgical Group 2007-2012
Established in 1948, the largest and oldest private academic plastic surgical practice in North America, voted by LI Press as the best plastic surgery practice in Long Island 2008-2012
More
- Director Clinical Research, NUMC Plastic Surgery Residency Program 2005-20013
- Partner, Long Island Plastic Surgical Group 2000-2013
- Course Director, Fresh Cadaver Plastic Surgery Lab 2001-present
- Medical Advisory Board, HealthiNation 2009-present
- Governing Board Member , Global Medical Relief Fund 2008- present
- Chief Medical Officer, Klinger Advanced Aesthetics 2005-2007
- Vice-President, Long Island Plastic Surgical Group 2005-2007
- Chief of Microsurgery, Winthrop University Hospital 2003-2008
- Vice-Chairman, Plastic and Reconstructive Surgery Winthrop University Hospital 2003-2008
- Treasurer, New York State Society of Plastic and Reconstructive Surgeons, 2012-present
- Treasurer, New York Regional Society of Plastic and Reconstructive Surgeons, 2012-present
- Member at Large, Nassau Surgical Society, 2012-present
- Medical Advisor, Cytori Corp. 2012-present
- Committee Member, Plastic Surgery Educational Foundation 2000-present
- Medical Board Member, Klinger Advanced Aesthetics 2006-2007
- Membership Committee Director, Long Island Chapter, 2005-present Northeastern Society of Plastic and Reconstructive Surgeons
- American Society of Plastic Surgeons
- Research and Technology Committee Member, 2011-present
- Public Education Committee Member, 2005-2011
- Clinical Trials Committee Member, 2009-2011
- Malpractice crisis task force 2008-2010
- Health Policy Committee Member, 2007-2008
- Quality and Performance Measurement 2007-2008
- American Society Aesthetic Plastic Surgeons
- Practice Relations Committee Member 2007-present
- Associate Program Director, Plastic Surgery Residency Program, Nassau University Medical Center 2001-2006
- Best Teaching Award 2003
- National Trainer, Sculptra Aesthetic 2009- present
- Medical Advisor, Life Cell Corporation 2008- present
- Medical Advisor, Kensey Nash 2010-present
- Medical Advisor, Mentor/Ethicon 2008- present
- Course Director, Klinger Advanced Aesthetics 2005-2006 Aesthetic Training Program Award
- Medical Advisory Board, Happy Face Foundation 2001-2004